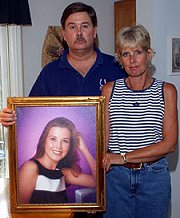
Gary and Ruth Pettijohn, of Batesville, Ind., hold a photo of their daughter, Kristen, who died after being prescribed a powerful antibiotic called Avelox. RANDY AMICK, KRT
The drug Kristen Pettijohn took was called Avelox. It's part of a family of
antibiotics called fluoroquinolones.
Those powerful but risky drugs are intended for patients who are fighting
particular bacterial bugs. But they're widely prescribed off-label for less
serious illnesses, sometimes even to treat viruses, which can't be killed by
antibiotics.
A study this year funded by the National Institutes of Health reviewed 100
emergency room prescriptions for fluoroquinolones and found that only 19
were written for appropriate conditions and only one was given in the
correct dose and for the proper duration.
The FDA long has been aware of the possibility that Avelox could be misused.
Just before it approved Avelox in 1999, a member of the agency's expert
review panel — Robert Danner, a critical care expert at the NIH — offered a
warning: "This is exactly the kind of place that you get into trouble. … I
am absolutely convinced that the drug will be used differently once it's
marketed frequently."
Avelox was approved, however, and marketed hard by Bayer Corp. In 1999 and
2001, the FDA admonished company officials for encouraging unapproved uses.
This past May, Pettijohn, a gregarious 23-year-old nursing student from
Batesville, Ind., who recently had gotten engaged, picked up the persistent
cold that had been running through her family. "Her version was a little
worse than ours," said her father, Gary Pettijohn. "I would say it was
moderate at best."
Early in the morning of May 15, Pettijohn's mother took her to an emergency
room. Going there, Pettijohn told her mom, would be quicker than waiting for
an appointment with their family physician.
Forty-two minutes later, Pettijohn was on her way to the drugstore. The
doctor had diagnosed her with acute bronchitis and prescribed Avelox. The
potent antibiotic's label says it's approved for cases of chronic, or
long-term, bronchitis, and only after blood tests have been taken to
identify the bacteria causing the problem. Her medical records show no blood
work was done.
That was a Thursday. By Sunday, Pettijohn was nauseated and suffering
abdominal pain. Her mother packed a plastic bag with the remaining Avelox
pills and took her to the hospital.
Over the next five days, Pettijohn was incoherent. She had a burning rash
and her skin began peeling off. She slipped into a coma, resting on an air
bed, totally wrapped as though she were a severe burn patient.
By Wednesday, a doctor approached Gary and Ruth Pettijohn.
"Our problem just got twice as difficult," he said. "She has two
life-threatening conditions simultaneously."
Pettijohn's liver was in full failure, and she was experiencing a form of
Stevens-Johnson syndrome, a rare and extreme drug reaction mentioned on the
Avelox label.
She had a liver transplant on Friday. The doctors reported that her old
liver had turned to mush and fallen apart in their hands.
Soon after the operation, Pettijohn had a heart attack, then another. Her
death certificate cited Avelox as the prime contributing factor in her
death.
The hospital had no comment about her death. Bayer had no comment beyond
saying the death "was promptly and accurately reported to the FDA," and that
it thinks its antibiotic should be prescribed only for approved conditions.